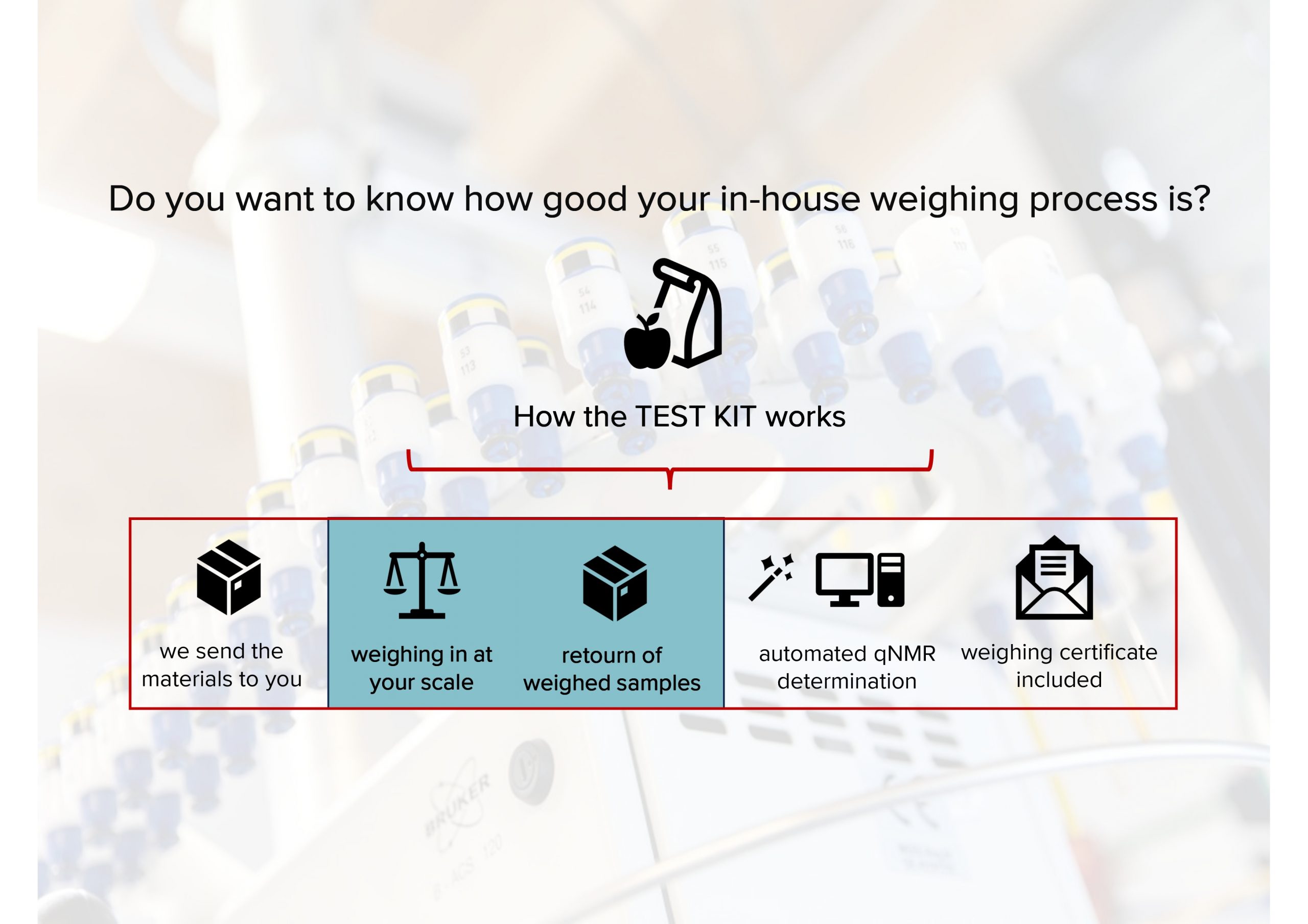
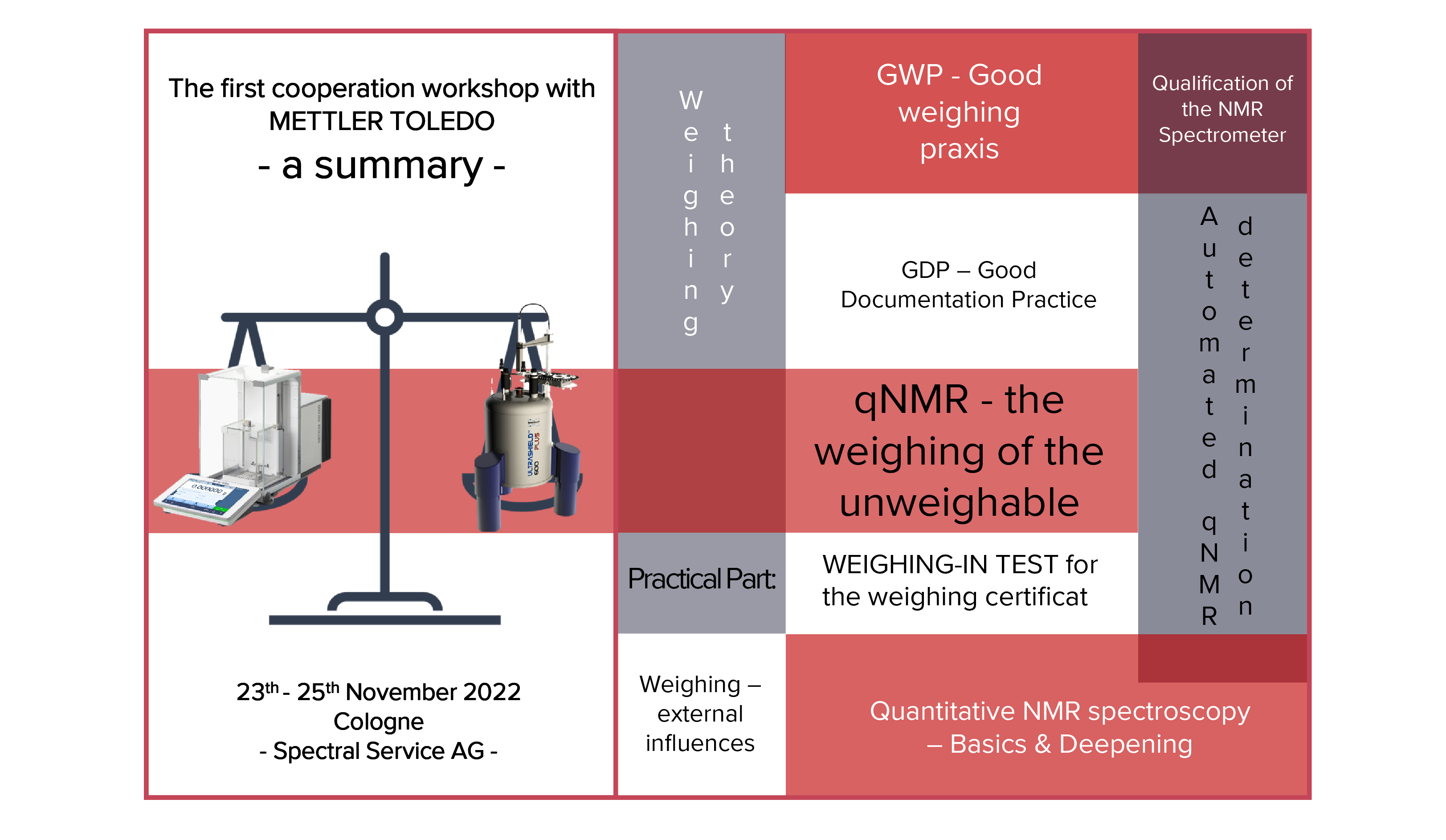
Since our last GLP (Good Laboratory Practice) inspection was 3 years ago, we recently contacted the authorities for recertification. Now, we have it concrete:
The pre-inspection will take place in July 2018 and the main inspection will be proceeded at the end of August 2018.
We are looking confidently forward to the inspection and are happy to welcome the inspectors at our testing facility.
Just to remember – what is behind the system?
According to OECD, Section I, art. 2.1.1. Good Laboratory Practice (GLP) is a quality system concerned with the organisational process and the conditions under which non-clinical health and environmental safety studies are planned, performed, monitored, recorded, archived and reported.
It is applicable for analytical and research laboratories to ensure the uniformity, consistency, reliability, reproducibility, quality, and integrity of chemical non-clinical safety tests; e.g. from physio-chemical properties through acute to chronic toxicity tests.
In Germany, GLP is based on the German Chemicals Act (Deutsches Chemikaliengesetz).
Through the worldwide implementation and broad mutual recognition of test data, this quality assurance system has contributed to health and environment protection and also to animal protection like no other system before.