GECA Pharma GmbH (Johannes Gallois): Import and distribution of cannabis in Germany
Since March 2017, medical cannabis can be prescribed as a medical drug in Germany. Patient numbers have increased from 1.200 to more than 40.000 within the first 18 months. Experts estimate a potential of up to 1.000.000 patients in Germany. Globally changing regulations lead to a fast-growing new industry. Medical benefits are now in the spotlight and corporations seek global strategic partnerships. These partnerships aim at bringing together producers, importers, distributors and research institutes for quality control and analysis in order to set industry standards. GECA Pharma is one of the first cannabis importers and distributors in Germany. GECA holds the pharmaceutical wholesale license and the permit to handle narcotics. Imports are strictly regulated and supervised by the Federal Institute for Drugs and Medical Devices. Domestic affairs like the tendering process for cultivation are controlled by the German Cannabis Agency, imports are regulated by the Federal Institute for Narcotics (Bundesopiumstelle). Importers need the required permissions, infrastructure and qualified persons to handle narcotics. Transport, warehousing and distribution need to be GDP certified and all processes need to be documented completely. Quality and security controls are crucial throughout the entire value chain. Medical cannabis can only be imported from GMP certified producers and all transports need to be GDP compliant. The distribution can only be handled through pharmacies which have the necessary license for narcotics. Prior to handing out the medicine to the patient, the pharmacy needs to check the narcotic prescription and realize an identity verification of the substance.
Steelyard Analytics, Inc. (Kristie Adams): Reefer Madness: The Curious Case of Cannabis in America

Mestrelab Research S.L. (Niccy Tonge): Targeted NMR Mixture Analysis: meeting the throughput challenge
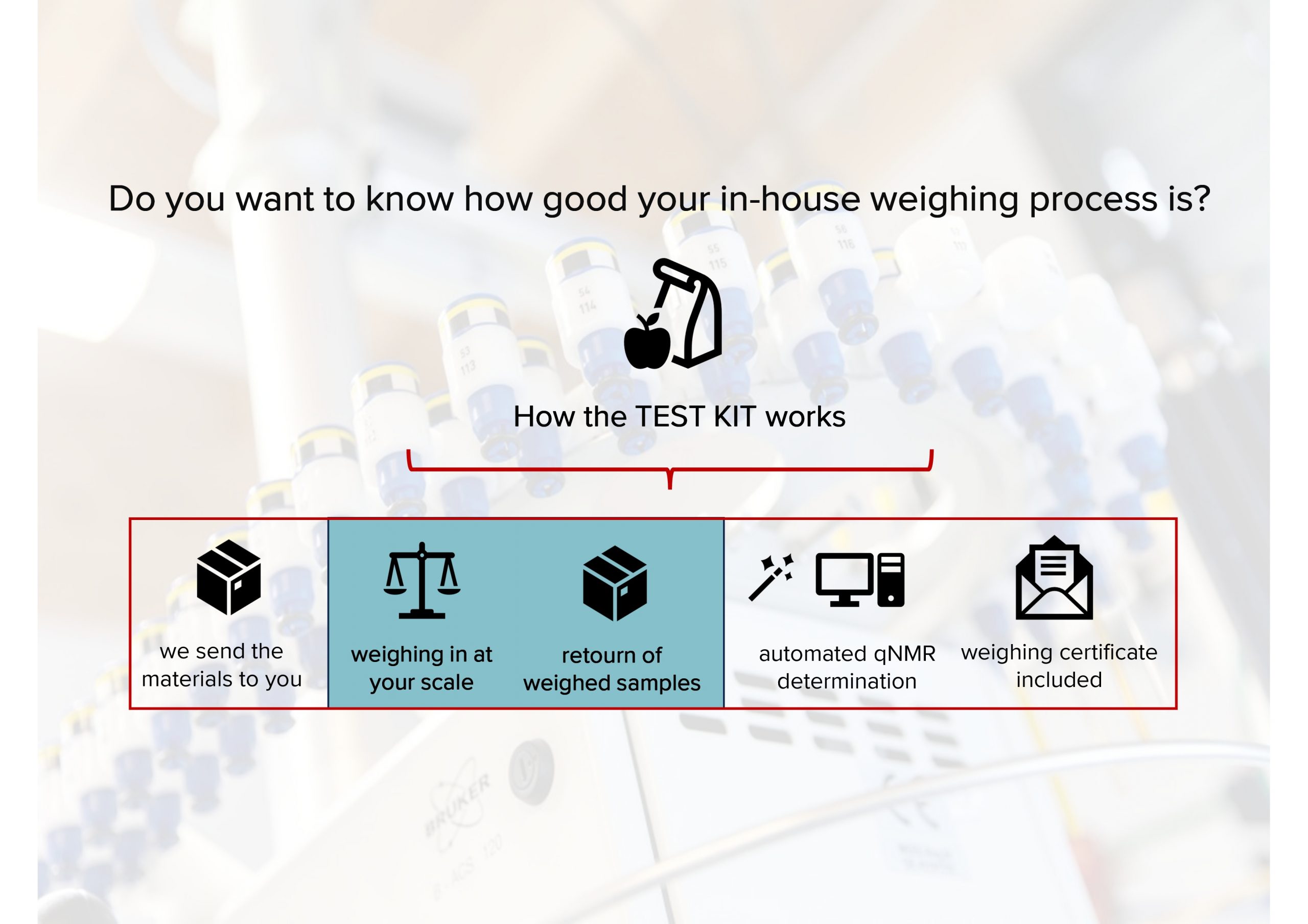
Mestrelab started the presentation by explaining the products and services, and identifying how they fit with the analysis of cannabis products. An overview of the software capabilities was presented, highlighting specific features which are directly relevant to cannabis analysis. Next the focus was shifted to targeted NMR analysis of cannabis, highlighting the other analytical techniques which can be used and identifying why these methods have drawbacks and disadvantages. Finally, the results of an analysis was presented, highlighting the potential for using NMR for this type of analysis and demonstrated that when it is coupled with the correct automation, NMR can provide rapid and reliable results for the identification of compounds in a cannabis sample with essentially no user interventions and in a fully quantitative manner.
Verity Analytics (Andrew Lewis): The Good Oil on Cannabis Oils
Oils, derivatives and edibles are a huge part of the recreational and medical cannabis market which is growing very quickly worldwide. Oils and extracts are made using a wide variety of techniques including immersion in oils, fats, butter, and ice water, or ethanol, supercritical CO2, hexane or butane extraction. The resulting oils and waxes obtained by extraction have elevated concentrations of actives like CBD (if hemp used) or THC (cannabis): dried plant 5-20 wt % , oils waxes, 30-90 wt %, shatter up to 97 wt%. 1H and 13C qNMR can be used to easily distinguish and quantify the main cannabinoids of interest THC, THCA, CBD, CBN, CBG etc. and various deuterated solvents can be used, resonances have different chemical shifts in different solvents. NMR potentially faster and cheaper than other techniques: simpler sample preparation, shorter measurement time, and requires less solvent, plus requirement for daily calibration using expensive reference standards. There are currently only a limited number of cannabioid refecence standards aveilable commercially and they usually come as dilute (1 mg / mL) solutions in non-deuterated solvents. Other analytical techniques like GC, GC-MS, LC-MS, HPLC, and convergence chromatography typically used for detecting and quantifying cannabinnoids already have stadard methods published e.g. by instrument manufacturers, but NMR has advantages over these, especially when they are in complex matrices like skin creams & edibles (example shown was cookies and brownies dissolved in MeOD with sample preparation and analyisis time < 20 min by NMR versus 1 day by GC-MS). NMR can also detect presence of, and quantify, other compounds like lipids, sugars, residual solvents, water, other unwanted compounds (e.g. synthetic cannabinoid mimics). Several groups have or are working on) publishing standard methods for cannabis ID and analysis including AOCS, UPS, AHP, ASTM.