What is GMP?
GMP refers to the Good Manufacturing Practice regulations released by the US Food and Drug Administration (FDA), International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH) and the European Pharmacopoeia (Ph. Eur.). The aim of GMP is to guarantee human safety by ensuring that patients receive pharmaceutical products in high quality. And this quality requirements have to be met during all stages of the product: manufacture, processing, packaging and storage of pharmaceuticals as well as the chemical, physical and biological analysis. To obtain a license for the retail of pharmaceutical products companies all over the world must comply with the GMP regulations.
Why is GMP important?
GMP regulations have been introduced to guarantee safety, effectiveness, uniformity, and to minimize or even eliminate mistakes, failures, as well as health risks for manufacturer and consumer.
Should my sample be analyzed under GMP?
It is necessary to categorize the type of sample: is it a (novel) food product, or a supplement, does it belong to cosmetics, or is it a chemical or something completely different? In the case of a cosmetic or pharmaceutical product, the probability of a required NMR analysis under GMP is very high. For the NMR analysis of food products and chemicals, a proceeding under GMP is not required. However, we recommend gathering information about the postulated regulations for the individual sample.
NMR Analysis under GMP
Spectral Service AG provides GMP compliant NMR analysis as pharmaceutical analysis contract services. We support you from early structure elucidation, product development, stability testing, NMR method development and validation through to routine batch testing.
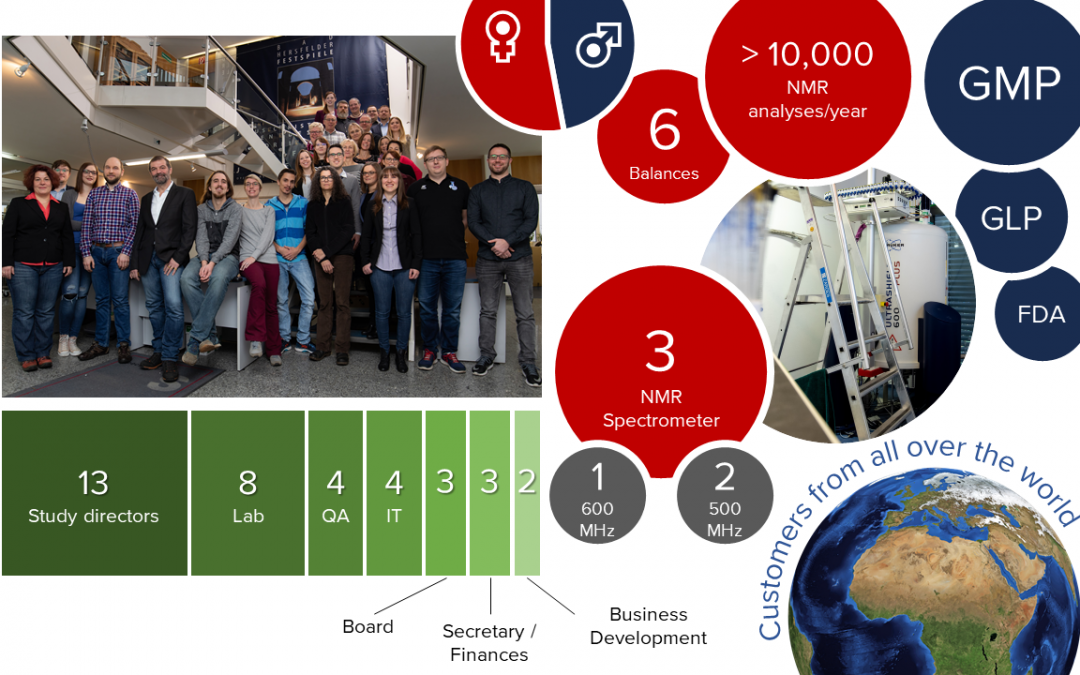
Our analytical scientists are highly trained professionals with years of experience in method development, validation and NMR analysis of different kinds of products. Our team of 35 people is committed to the success of our clients – of you!